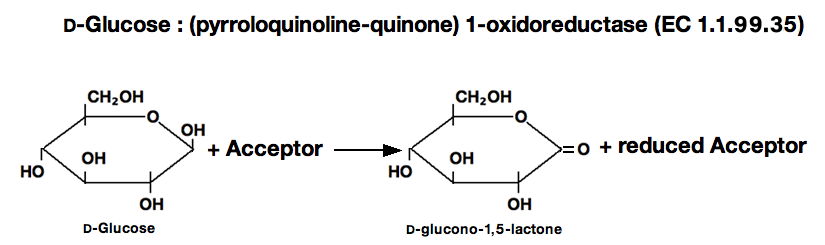
GLUCOSE DEHYDROGENASE (PQQ-dependent) from Microorganism
GLD-321
| Appearance: | Purple amorphous powder, lyophilized | |
|---|---|---|
| Activity: | GradeⅢ 500U/mg-solid or more | |
| Contaminants: | Glucose dehydrogenase ≤1.0×10⁻³% (NAD-dependent) Hexokinase ≤1.0×10⁻³% |
|
| Stabilizers: | Ca⁺⁺, BSA | |
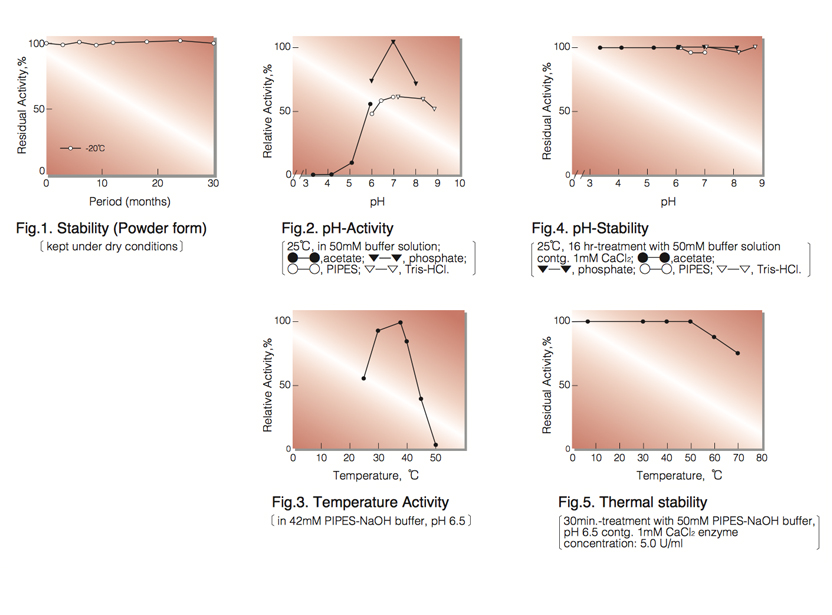
| Stability: | Stable at -20°C for at least One year (Fig.1) |
|---|---|
| Molecular weight: | approx. 100,000 (by gel filtration) |
| Michaelis constant: | 4.8mM (D-Glucose) |
| Inhibitors: | Cu⁺⁺, Pb⁺⁺, Ag⁺ |
| Optimum pH: | 7.0(Fig.2) |
| Optimum temperature: | 37℃(Fig.3) |
| pH Stability: | pH 3.5-8.5 (25℃, 16hr)(Fig.4) |
| Thermal stability: | below 50°C (pH 7.5, 30min)(Fig.5) |
| Substrate specificty: | (Table 1) |
| Effect of various chemicals: | (Table 2) |
APPLICATIONS
This enzyme is useful for enzymatic determination of D-Glucose.
ASSAY
Principle:
PQQ-glucose dehydrogenase
D-glucose+PMS ►D-glucono-1, 5-lactone+PMS (red)
2PMS (red)+NTB ►2PMS+Diformazan
The appearance of diformazan formed by the reduction of nitrotetrazorium blue (NTB) with phenazine methosulfate (PMS)(red) is measured at 570nm by spectrophotometry.
Unit definition:
One unit causes the formation of one half micromole of diformazan per minute under the conditions described below.
Method:
| A. D-Glucose solution: | 1M [1.8g D-Glucose (MW=180.16)/10ml H₂O]keep this solution at room temperature at least 3 hours before use |
|---|---|
| B. PIPES-NaOH buffer, pH 6.5: | 50mM [Weight 1.51g of PIPES (MW=302.36), suspended in 60ml of H₂O, dissolve with 5N NaOH and add 2.2ml of 10% Triton X-100. After adjusting pH to 6.5±0.05 at 25°C with 5N NaOH, fill up to 100ml with H₂O] |
| C. PMS solution: | 3.0mM [9.19mg Phenazine methosulfate (MW=306.34)/10ml H₂O] |
| D. NTB solution: | 6.6mM [53.96mg nitrotetrazorium blue (MW=817.65)/10ml H₂O] |
| E. Enzyme diluent: | 50mM PIPES-NaOH buffer, pH 6.5 containing 1mM CaCl₂, 0.1% Triton X-100, 0.1% BSA |
Procedure
| Concentration in assay mixture | |
|---|---|
| PIPES-buffer | 42 mM |
| D-Glucose | 30 mM |
| PMS | 0.20mM |
| NTB | 0.22mM |
1. Prepare the following reaction mixture in a brownish bottle and store on ice. (Prepare freshly)
0.9 ml D-Glucose solution (A)
25.5ml PIPES-NaOH buffer, pH 6.5 (B)
2.0ml PMS solution (C)
1.0ml NTB solution (D)
2. Pipet 3.0ml of working solution into a test tube (plastic tube) and equilibrate at 37°C for about 5 minutes.
3. Add 0.1ml of enzyme solution* and mix by gentle inversion.
4. Record the increase of optical density at 570nm against water for 4 to 5 minutes in a spectrophotometer
thermostated at 37°C, and calculate the ΔOD per minute from the initial linear portion of the curve (ΔOD test).
At the same time, measure the blank rate (ΔOD blank) by the same method as test except that the enzyme diluent (E) is added instead of the enzyme solution.
* Dissolve the enzyme preparation on ice cold enzyme diluent (E) and dilute to 0.1-0.8U/ml with the same buffer, immediately before assay. (The use of plastic tube is recommended because of sticky nature.)
Calculation
Activity can be calculated by using the following formula :

ΔOD/min (ΔOD test−ΔOD blank ) ×Vt × df
Volume activity (U/ml) = =ΔOD×1.54×df
20.1×1.0×Vs
Weight activity (U/mg)=(U/ml)×1/C
- Vt
- : Total volume (3.1ml)
- Vs
- : Sample volume (0.1ml)
- 20.1
- : Half a millimolar extinction coefficient of diformazan (㎠/0.5micromole)
- 1.0
- : Light path length (cm)
- df
- : Dilution factor
- C
- : Enzyme concentration in dissolution (c mg/ml)
REFERENCES
- K.Matsushita et al.; FEMS Microbiology Letters, 55, 53 (1988).
| Substrate (50mM) | Relative activity(%) | Substrate (50mM) | Relative activity(%) |
|---|---|---|---|
| D-Glucose | 100 | Galactose | 16.0 |
| L-Glucose | 0.3 | D-Lactose | 68.9 |
| D-Xylose | 15.0 | D-Sorbitole | 0.2 |
| 2-Deoxy-glucose | 4.9 | D-Mannitol | 0.0 |
| L-Sorbose | 0.5 | Sucrose | 0.2 |
| D-Mannose | 10.8 | Inositol | 0.0 |
| D-Fructose | 0.3 | Maltose | 107.0 |
| Chemical | Concn.(mM) | Residual activity(%) |
Chemical | Concn.(mM) | Residual activity(%) |
|---|---|---|---|---|---|
| None | − | 100 | MIA | 2.0 | 87 |
| Metal salt | 2.0 | NEM | 2.0 | 100 | |
| MgSO₄ |
108 | IAA | 2.0 | 98 | |
| CaCl₂ | 108 | Hydroxylamine | 2.0 | 19 | |
| Ba(OAc)₂ | 105 |
EDTA | 5.0 | 79 | |
| FeCl₃ | 79 |
o-Phenanthroline | 2.0 | 7 | |
| CoCl₂ | 42 | α,α′-Dipyridy | 1.0 | 103 | |
| MnCl₂ | 105 | Borate | 5.0 | 110 | |
| ZnCl₂ | 45 | NAF | 2.0 | 111 | |
| Cd(OAc)₂ | 107 | NaN₃ | 2.0 | 115 | |
| NiCl₂ | 101 | Triton X-100 | 0.10% | 101 | |
| CuSO₄ | 0 | Brij 35 | 0.10% | 22 | |
| Pb(OAc)₂ | 0 | Tween 20 | 0.10% | 104 | |
| AgNO₃ | 0 | Span 20 | 0.10% | 60 | |
| HgCl₂ | 77 | Na-Cholate | 0.10% | 67 | |
| 2-Mercaptoethanol | 2.0 | 99 | SDS |
0.05% | 33 |
| PCMB | 1.0 | 97 | DAC | 0.05% | 113 |
Ac, CH₃CO; PCMB, p-Chloromercuribenzoate; MIA, Monoiodoacetate; NEM, N-Ethylmaleimide; IAA, Iodoacetamide; EDTA, Ethylenediaminetetraacetate; SDS, Sodium dodecyl sulfate; DAC, Dimethylbenzyl-alkyl-ammonium chloride.